Relu Secures FDA 510(k) and CE Marking for Revolutionary Dental Software Relu® Creator
Comunicato Precedente

Comunicato Successivo

FDA clearance signifies compliance with U.S. medical device standards, while the CE Mark confirms adherence to EU regulations for product safety and consumer protection. This dual recognition underscores Relu's commitment to excellence in the oral health sector.
"We are thrilled to have attained both FDA and CE approval for the Relu® Creator," stated Adriaan Van Gerven, CTO and Co-founder, of Relu. "These significant achievements are a testament to Relu's dedication to creating superior technology that meets the highest international safety and quality standards. We're poised to revolutionize patient and dentist experiences across both continents."
"This approval paves the way for our advanced technology to enter US and EU dental markets with the highest potential for growth. Having led strategy in Dental, AI and Orthodontic sectors, I consider this achievement a key differentiator that sets Relu® Creator apart. This is a major step forward in Relu's mission to modernize dental care," said Bindu Saran, former Global Head of Orthodontic Technology at Straumann, former Sr. Manager of R&D at Ormco, and Board Member at Relu.
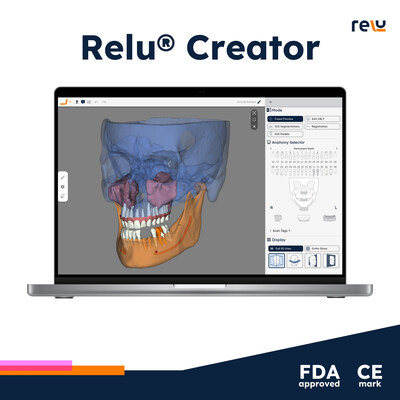
The Relu® Creator integrates cutting-edge AI to deliver features such as automated anatomical identification, and enhanced 3D imaging for dental diagnostics and treatment planning. By improving precision and increasing overall efficiency in dental procedures, Relu® Creator is set to be an essential tool for modern dental clinics.
About Relu
Relu is founded in 2019 with the dream of making dental treatments safer and faster. They plug in advanced computer vision and artificial intelligence to automate manual workflows. Their Relu® Engine and Relu® Creator are used by dental lab and software partners for more than thousands of orthodontic and implant treatments every day. Relu is headquartered in Leuven, BE with an office in Boston, MA, USA. Learn more at relu.eu.
© 2024 Relu
![]() View original content:https://www.prnewswire.co.uk/news-releases/relu-secures-fda-510k-and-ce-marking-for-revolutionary-dental-software-relu-creator-302246365.html
View original content:https://www.prnewswire.co.uk/news-releases/relu-secures-fda-510k-and-ce-marking-for-revolutionary-dental-software-relu-creator-302246365.html





